- Published:
- Last Updated: July 26th, 2023

Attorney Sean T. Keith has been a personal injury lawyer for 30+ years, a nationally recognized Top 100 Trial Lawyer, and top car accident lawyer & motor vehicle accident lawyer in Arkansas.
Legally Reviewed
This article has been written and reviewed for legal accuracy and clarity by the team of writers and attorneys at Keith Law Group and is as accurate as possible. This content should not be taken as legal advice from an attorney. If you would like to learn more about our owner and experienced injury lawyer, Sean T. Keith, you can do so here.
Fact-Checked
Keith Law Group does everything possible to make sure the information in this article is up to date and accurate. If you need specific legal advice about your case, contact us. This article should not be taken as advice from an attorney.
Sean Keith's Accollades & Practice Areas He Specializes In
- Over $20 Million recovered in Medical Device Injury Lawsuits.
- Over $13 Million recovered in Car Accident Lawsuits and other Motor Vehicle Accident Lawsuits.
- Over $100 Million recovered in total on behalf of clients.
- Sean represents clients in cases involving personal injuries, car accidents, motorcycle accidents, truck accidents, wrongful death, slip and falls, nursing home abuse cases, nursing home elopement cases, and more.
A Guide To Arkansas Hernia Mesh Injuries
According to the FDA, more than one million hernia repairs are performed each year in the U.S. Hernia meshes are used by surgeons frequently.
Usually, with various types of surgical treatments, the patient recovers.
However, patients may be left with hernia mesh injuries due to complications from a defective mesh product.
The defective mesh products can cause a wide range of injuries such as organ damage, intestinal and bowel obstructions, adhesions, infections, and permanent nerve damage.
They also cause other adverse side effects such as chronic pain, inflammation, hernia recurrence, and the need for additional or revision surgery to remove the mesh and rectify the problem.
If you have developed an illness or health complication as a result of using any hernia mesh medical device, you may be entitled to compensation.
Our attorneys at Keith Law provide dedicated, experienced legal representation to victims of hernia mesh injuries.
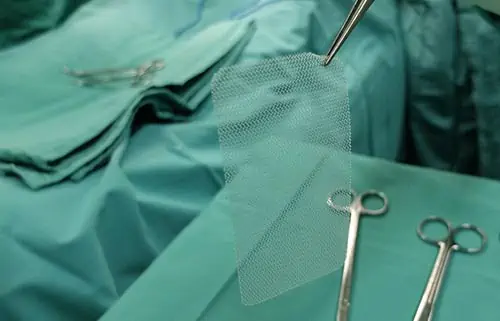
Table of Contents
What Is a Hernia and How Can It Be Treated?
A hernia occurs when an organ, intestine, or fatty tissue squeezes through a hole or a weak spot in the surrounding muscle or connective tissue.
They commonly occur in the abdomen.
According to the FDA, the most common types of hernias include:
- Inguinal: occurs in the inner groin
- Femoral: occurs in the upper thigh/outer groin
- Incisional: occurs through an incision or scar in the abdomen
- Ventral: occurs in the general abdominal/ventral wall
- Umbilical: occurs at the belly button
- Hiatal: occurs inside the abdomen, along with the upper stomach/diaphragm
Although surgery is the only treatment that can repair hernias, waiting and watching is an option for many people who don’t have complications or symptoms with an adult inguinal hernia.
In laparoscopic hernia repair, the surgeon makes several small incisions in the abdomen.
Open repair involves a larger incision.
Both laparoscopic and open repair can be performed with or without mesh.
Symptoms of Hernia Mesh Injuries
Common symptoms of hernia mesh injuries are bloating, bulging, or bowel obstruction.
The patient might feel heavy underneath their stomach.
At times, they won’t be able to lift heavy things.
They might feel exhausted even after doing nothing.
If you have a hernia mesh, some of the symptoms you can be on the lookout for are urinary issues, severe pain, irritable bowel, fever, fatigue, and constipation.
These symptoms can occur within days or years after the mesh was installed.
If you experience any of these symptoms, contact your physician immediately.
Have There Been Any Hernia Mesh Recalls?
Numerous hernia mesh products have been recalled over the years.
In 2005, C.R. Bard recalled various Kugel Mesh branded products after they were associated with numerous health complications like increased risk of bowel tears.
In 2012, Atrium was given a warning by the FDA for failing to recall its mesh products named C-QUR.
In 2015, the company was instructed by a federal judge to stop manufacturing the subject product as a result of numerous complaints of infections from the public.
In 2016, Ethicon undertook a voluntary recall of its Physiomesh products after studies showed that its products were defective.
The company was forced to issue notices to doctors and hospitals informing them to stop using the subject products as they increased the chances of hernia failure and other serious complications.
Professional Help Can Guide You Better
Hernia mesh medical devices have resulted in thousands of patients suffering from serious adverse health complications such as infections and organ damage.
Injuries related to this device can be avoided by using an alternative.
However, if you or your loved ones have suffered hernia mesh injuries in Arkansas, reach out to us, so we can guide you through the best options available to you.
We are experienced injury attorneys in Rogers and we are here to help you.
Practice Areas
You pay
Nothing
unless we win
Do You Have A Case?
Recent Legal Posts & Articles
Recent Legal Guides

Choose Us For Your Personal Injury Case
- Available 24/7
- No Upfront Fees
- Free Case Evaluation
- No Fees Unless We Win!















